On the topic of trending and/or newer compounds that have taken the supplement industry by storm, sulforophane (SFN) has stood out over recent years. Coming from the Brassica family of vegetables, this isothiocyanate has gained popularity as a nutraceutical to improve health and as a pharmaceutical to treat several diseases. On top of being significantly more bioavailable than curcumin (20-fold higher) and quercetin (80-fold higher), two of the most common supplement antioxidants, sulforophane has shown promise in several areas of medical research, including cancer treatments, metabolic disorders, and neurological disorders, amongst others (Mahn & Castillo, 2021). Here are the main benefits and treatment uses of sulforophane supplementation.
Mechanisms of action and detoxification
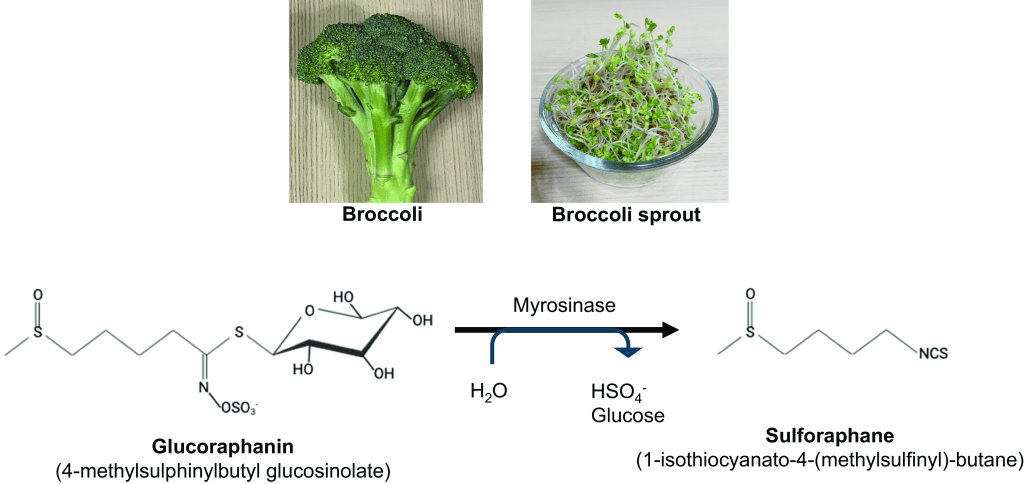
Sulforophane is formed through the hydrolysis of glucoraphanin, which is found in cruciferous (Brassica family) vegetables, such as kale, broccoli, cabbage, kale and cauliflower, by the enzyme tyrosinase. Sulforophane is known to be a potent inducer of phase II detoxification enzymes and the same pathways that are triggered to induce these enzymes also up-regulate a series of key enzymes, such as NAD(P)H quinone dehydrogenase 1 (NQO1), heme oxygenase 1 (HO-1), quinone reductase, glutathione S-transferases (GST), and inducible nitric oxide synthase. Up-regulation of these enzymes confirm SFN’s potent antioxidant and anti-inflammatory activity (Saito et al, 2025).
Cancer
Sulforophane’s anti-cancerous effects are tied to it’s epigenetic modulation activity, which induce various cellular mechanisms that attack and/or prevent cancer cells from progressing, which include apoptosis activation, nuclear factor-κB (NF-kB) pathway inhibition, and cell cycle arrest induction (Hyun, 2020). Additionally, SFN’s immune-enhancing effects could further attest to SFN’s anti-cancerous effects, as research conducted on prostrate cancer showed an increase in natural killer cell cytotoxicity and T cell infiltration, resulting in a reduction of metastasis (Mahn & Castillo, 2021).
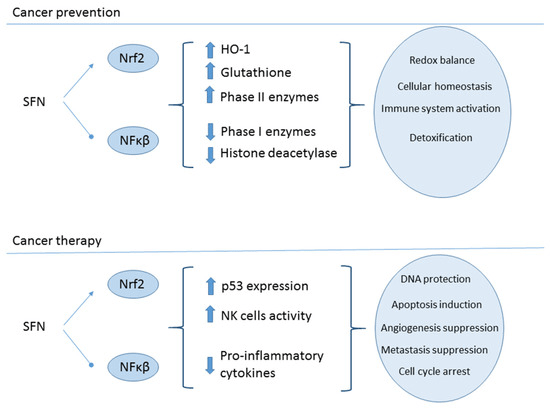
The bulk of research on isothiocyanates into cancer focuses on prostate, colon, and breast cancers. However, to outline the key anti-cancerous mechanisms in simple terms; molecular pathways, such as ERK1/2, MAPK, and JNK, instruct cells to grow and divide but isothicyanates will either down-regulate or deactivate them, preventing or reversing cancer cell growth. Additionally, chronic inflammation helps cancer cells grow and isothiocyanates can suppress inflammation fire starters such as cyclooxygenase-2 (COX-2), hindering tumour growth. If cancers can metastasise, then preventing the spread of cancerous cells into surrounding tissues should be paramount in cancer treatments and isothiocyanates could pose as useful treatment adjuncts, as they can reduce key molecules that cancers use to invade other tissues, such as matrix metallopeptidase-9 (MMP-9). And finally, key regulators of cell survival, inflammation and growth, such as NF-kB and AP-1, get blocked from entering the nucleus of cancer cells, which results in cancerous cells losing key survival signals (Soundararajan & Kim, 2018).
Metabolic health
Recall that sulforophane and other isothiocyanates tackle chronic illnesses by attenuating or preventing cellular mechanisms that drive them. In regards to diabetes and metabolic syndrome, there several key mechanisms that SFN targets, which in turn, improve treatment outcomes. The first one begins with SFN’s main cellular action, which is activating the Nrf2 pathway, the master switch for the body’s defence system. By doing so, SFN boosts antioxidant production, increases detoxification enzymes and reduces chronic inflammation, which improves insulin sensitivity, metabolic balance, and reduce cellular damage. Another pathway, NF-E2, closely linked to Nrf2, also enhances protective gene activity and reduces oxidative stress, a major player in metabolic syndrome. Additionally, by up-regulating insulin receptor substrate 1 (IRS-1), sulforophane improves insulin signalling, enabling cells to absorb glucose more effectively and in turn, lower blood sugar levels (Masoumvand et al, 2025).
In research conducted on mice, SFN attenuates weight gain induced by a high glycemic index diet, as well as significant improvements in glucose/insulin tolerance, insulin sensitivity(as eluded to above), and pancreatic b-cell function. It is evident that a high glycemic index diet, genes involved in energy metabolism get disrupted, which drives fat storage and metabolic dysfunction. In the same study, SFN supplementation up-regulated the PGC-1a gene, improving energy use and mitochondria production. Furthermore, SFN restored sirtuins (SIRT1, SIRT2, SIRT3), genes involved in regulating metabolism, improving metabolism and fat handling (Çubuk et al, 2026). Research conducted on obese/type 2 diabetic humans seems to allude to the same conclusions in rat studies. In fact, supplementing with broccoli sprout extract (a source of sulforophane) seemed to work better in obese patients with poorly controlled type 2 diabetes, in contrast to those with lower BMI. Statistically significant short and long-term improvements occurred in both fasting glucose and HbA1c (glycated haemoglobin) (Axelsson et al, 2017).
Neurological disorders
The precise molecular mechanisms behind acute and chronic neurodegenerative conditions isn’t concrete but there are several mechanisms, such as oxidative stress, misfolding, aggregation, and accumulation of proteins, perturbed calcium homeostasis, excitotoxicity, inflammation, and apoptosis, which have been outlined as players in their pathogenesis. The brain is particularly vulnerable to the effects of oxidative stress, due to its high oxygen consumption and oxidisable polyunsaturated fatty acids content. As mentioned above, the body’s antioxidant defence systems are regulated by mechanisms that involve the proteins, nuclear factor erythroid 2-related factor 2 (Nrf2) and Kelch-like-ECH-associated protein 1 (Keap1). By directly raising glutathione and antioxidant enzyme levels, sulforophane can attenuate oxidative stress in both animal and human neuronal cell lines (Tarozzi et al, 2013). In the absence of Nrf2 transcriptional activity, the blood-brain barrier becomes compromised. SFN, via anti-inflammatory/apoptotic effects, can preserve tight junction alterations. Up-regulation of heme oxygenase-1 (HO-1) and Nrf2 activity in the brain is thought to be responsible for these effects. It has been noted in both animal and human research that depletion of brain glutathione exacerbates stroke infarct size and specifically in rodents, disrupts short-term spatial recognition, learning and memory and cognitive flexibility (Fahey et al, 2025).
Furthermore, as SFN exerts its anti-inflammatory effects, it reduces the neuronal damage mediated by microglial activation and regulating the levels of inflammatory mediators, such as tumor necrosis factor-α (TNF-α), interleukin (IL) 6, IL-1β, inducible nitric oxide synthetase (iNOS), and cyclooxygenase-2 (COX-2). Given that neurons are metabolically demanding cells, it should be paramount to protect mitochondria and through the Nrf2 pathway, it appears that sulforophane can promote the activation of genes that favor the mitochondrial biogenesis, thus preserving the mitochondrial complex I, II, and IV, to lead to the production of adenosine triphosphate (ATP), otherwise reduced by neuronal damage. In a review of mice research models with Alzheimer’s disease, Parkinson’s disease, and multiple sclerosis, the most consistent findings amongst all three conditions was reduction of oxidative stress, neuroinflammation, prevention of neuronal death (apoptosis)/(improved cell survivability), and improvements in cognitive and behavioural function (Schepici et al, 2020).
References
Axelsson, A.S., Tubbs, E., Mecham, B., Chacko, S., et al. (2017). Sulforaphane reduces hepatic glucose production and improves glucose control in patients with type 2 diabetes. Science translational medicine, 9(394). https://doi.org/10.1126/scitranslmed.aah4477
Çubuk, M., Pınar, A.A., Süleyman, B., & Taş, N.G. (2026). Sulforaphane Against the Metabolic Consequences of a High-Glycemic-Index Diet: Protective and Therapeutic Mechanisms Associated with Obesity and Insulin Resistance. Nutrients, 18(4), 574. https://doi.org/10.3390/nu18040574
Fahey, J.W., Liu, H., Batt, H., Panjwani, A.A., & Tsuji, P. (2025). Sulforaphane and Brain Health: From Pathways of Action to Effects on Specific Disorders. Nutrients, 17(8), 1353. https://doi.org/10.3390/nu17081353
Hyun, T.K. (2020). A recent overview on sulforaphane as a dietary epigenetic modulator. EXCLI journal, 19, 131-134. https://doi.org/10.17179/excli2019-2039
Mahn, A., & Castillo, A. (2021). Potential of Sulforaphane as a Natural Immune System Enhancer: A Review. Molecules, 26(3), 752. https://doi.org/10.3390/molecules26030752
Masoumvand, M., Ramezani, E., Eshaghi Milasi, Y., et al. (2025). New horizons for promising influences of sulforaphane in the management of metabolic syndrome: a mechanistic review. Naunyn-Schmiedeberg’s Archives of Pharmacology, 398, 4933–4946. https://doi.org/10.1007/s00210-024-03706-3
Saito, A., Ishikawa, S., Yang, K., Sawa, A., & Ishizuka, K. (2025). Sulforaphane as a potential therapeutic agent: a comprehensive analysis of clinical trials and mechanistic insights. Journal of Nutritional Science, 14, e65. https://doi.org/10.1017/jns.2025.10033
Schepici, G., Bramanti, P., & Mazzon, E. (2020). Efficacy of Sulforaphane in Neurodegenerative Diseases. International Journal of Molecular Sciences, 21(22), 8637. https://doi.org/10.3390/ijms21228637
Soundararajan, P., & Kim, J. S. (2018). Anti-Carcinogenic Glucosinolates in Cruciferous Vegetables and Their Antagonistic Effects on Prevention of Cancers. Molecules, 23(11), 2983. https://doi.org/10.3390/molecules23112983
Tarozzi, A., Angeloni, C., Malaguti, M., Morroni, F., et al. (2013). Sulforophane as a potential against neurodegenerative diseases. Oxidative medicine and cellular longevity, 2013, 415078 . https://doi.org/10.1155/2013/415078

Leave a comment